  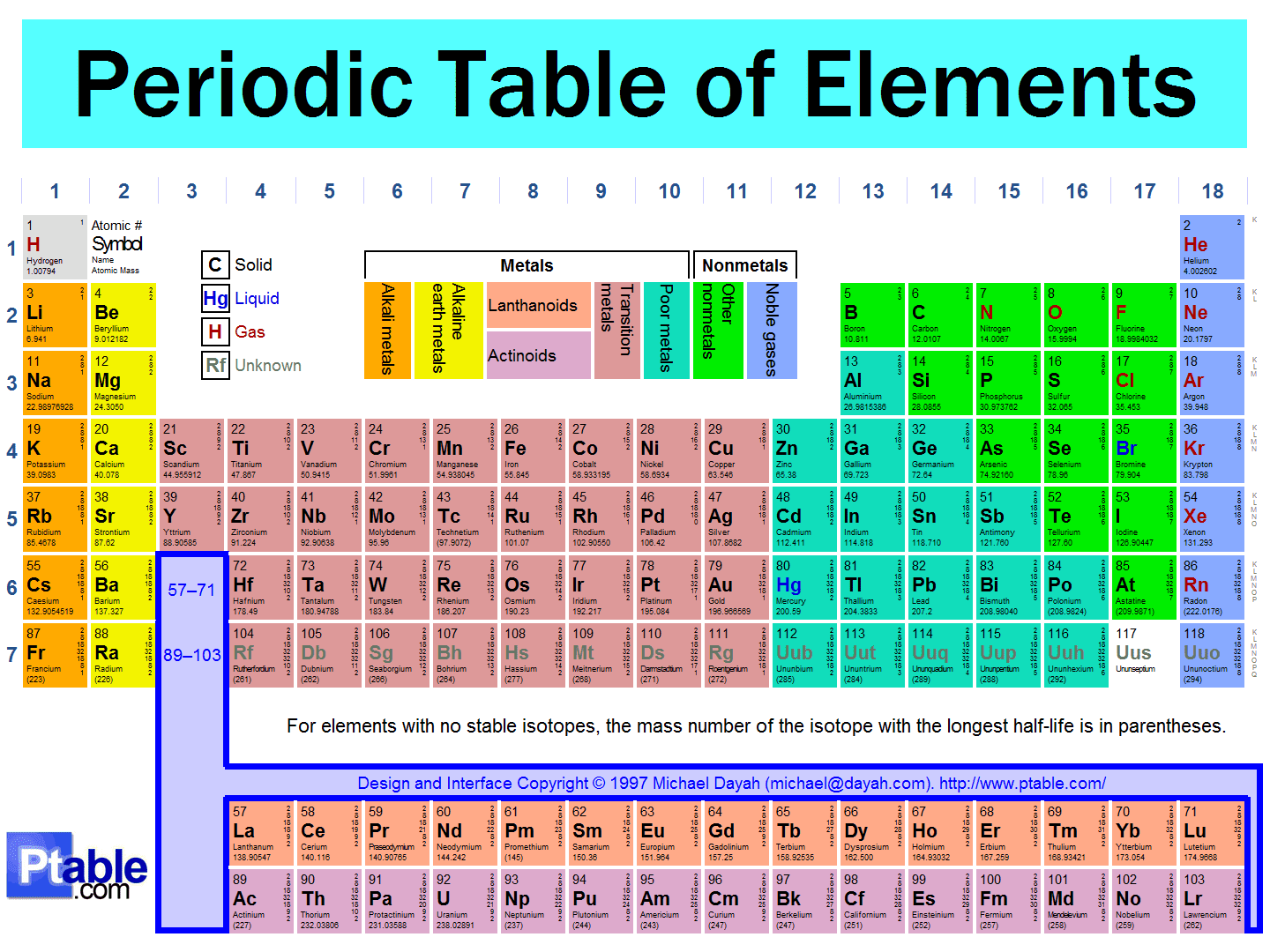
For example, all atoms with 6 protons are atoms of the chemical element carbon, and all atoms with 92 protons are atoms of the element uranium. The number of protons in an atom is called the atomic number. 2 Atoms are made up of protons, neutrons, and electrons. Ytterbium was demonstrated to be composed of ytterbium and a new element which was named scandium after Scandanavia, and finally ytterbium was split again to yield ytterbium and lutetium.Ī diagram showing the order in which the ten elements were isolated. A chemical element is a substance that is made up of only one type of atom. Holmium was then later demonstrated to be a mixture of holmium and dysprosium, which takes its name from the Greek word dysprositos meaning “difficult to get”, reflecting the difficulty found in isolating it. Terbium was later demonstrated to be a mixture of terbium and a new element which was named gadolinium in honour of Gadolin. Erbium was demonstrated to be a mixture of erbium and and a new element which was named ytterbium, again after the village of Ytterby.Įrbium was then itself demonstrated to a mixture of three elements: erbium thulium, named after Thule, a term used by early map makers for the far north where Sweden is located and holmium, named after the Swedish capital Stockholm. Get the facts about element Dysprosium (Dy) 66 from the periodic table. The original name was kept for one of these three parts and the other two elements named terbium and erbium, both after the village of Ytterby where they were found. In 1843 Carl Gustav Mosander demonstrated that ytterbite was actually made of three metal oxides, not one as Gadolin had thought. One of these chemists, Johan Gadolin, determined that ytterbite did indeed contain a previously unknown element and called this element yttrium. Naturally occurring dysprosium is composed of seven isotopes, the most abundant of which is 164Dy. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. Dysprosium is never found in nature as a free element, though it is found in various minerals, such as xenotime. 66 Dy Dysprosium 162.50 67 Ho Holmium 164.93 68 Er Erbium 167.26 69 Tm Thulium 168.93 70 Yb Ytterbium 173.05 71 Lu Lutetium 174.97 72 Hf Hafnium 178.49 73 Ta Tantalum 180.95. It is a rare earth element with a metallic silver luster. Click on the column header to sort the table by that column or click on an element name to get detailed facts about the element. Arrhenius took this ore, which he named “ytterbite”, and sent samples to various chemists for analysis. Dysprosium is a chemical element with symbol Dy and atomic number 66. Here's a list of all of the chemical elements of the periodic table ordered by increasing atomic number. The table below lists the atomic numbers, symbols, and names of all the elements, with the derivations for the symbols which are not of English origin. In 1787 one of the students of Lieutenant Carl Axel Arrhenius found a dark-coloured ore that was much too heavy to be coal. Atomic Names and Symbols Every element has a one- or two-letter abbreviation based either on its English name or its Latin or Greek name. This makes their extraction and isolation very difficult and this is where the “rare” in their name comes from. All of the elements are rare earth metals which occur in similar locations and have similar properties. The answer (and there is a tiny clue in some of the names) is that all ten elements were isolated from one sample, taken from a mine in the small village of Ytterby in Sweden. We created a timeline of the history of the periodic table.What do the following ten elements have in common? The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. Who invented the Periodic Table of Elements? The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. The Periodic Table of Elements can be used as an assisting tool in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. reaction with oxygen is slow at room temperature, but they can ignite around 150-200 C. How is the Periodic Table of Elements used? The Lanthanides have the following reactions: oxidize rapidly in moist air. 
The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. The Periodic table of elements is a tool, developed by scientists over hundreds of years. List of all the elements and their properties: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
